|
5/11/2024 0 Comments Co2 electron pair geometry chart
#6#: octahedron (a flat square with two "spokes") #5#: trigonal bipyramid (a trigonal planar shape with two "spokes") #3#: trigonal plane (a flat equilateral-triangle-looking shape) As it has a VSEPR shape #AX_5E_0# it is a trigonal bipyramid.Įach steric number has a same "basic shape": Its steric number is #5# due to the #5# bonded atoms to the central #S# atom plus #0# lone electron pairs. Thus, it is in the form #AX_3E_1#, which forms a trigonal pyramidal shape. #N#, the central atom, has a steric number of #4#, calculated by the #3# atoms it's bonding with #+1# lone pair. This is the total number of electron pairs and bonds with other atoms. Chem 14B Uploaded Files (Worksheets, etc.Find the central molecules' steric numbers.Arrhenius Equation, Activation Energies, Catalysts.Method of Initial Rates (To Determine n and k).Interesting Applications: Rechargeable Batteries (Cell Phones, Notebooks, Cars), Fuel Cells (Space Shuttle), Photovoltaic Cells (Solar Panels), Electrolysis, Rust.Appications of the Nernst Equation (e.g., Concentration Cells, Non-Standard Cell Potentials, Calculating Equilibrium Constants and pH).Work, Gibbs Free Energy, Cell (Redox) Potentials.Galvanic/Voltaic Cells, Calculating Standard Cell Potentials, Cell Diagrams.Biological Examples (*DNA Structural Transitions, etc.). 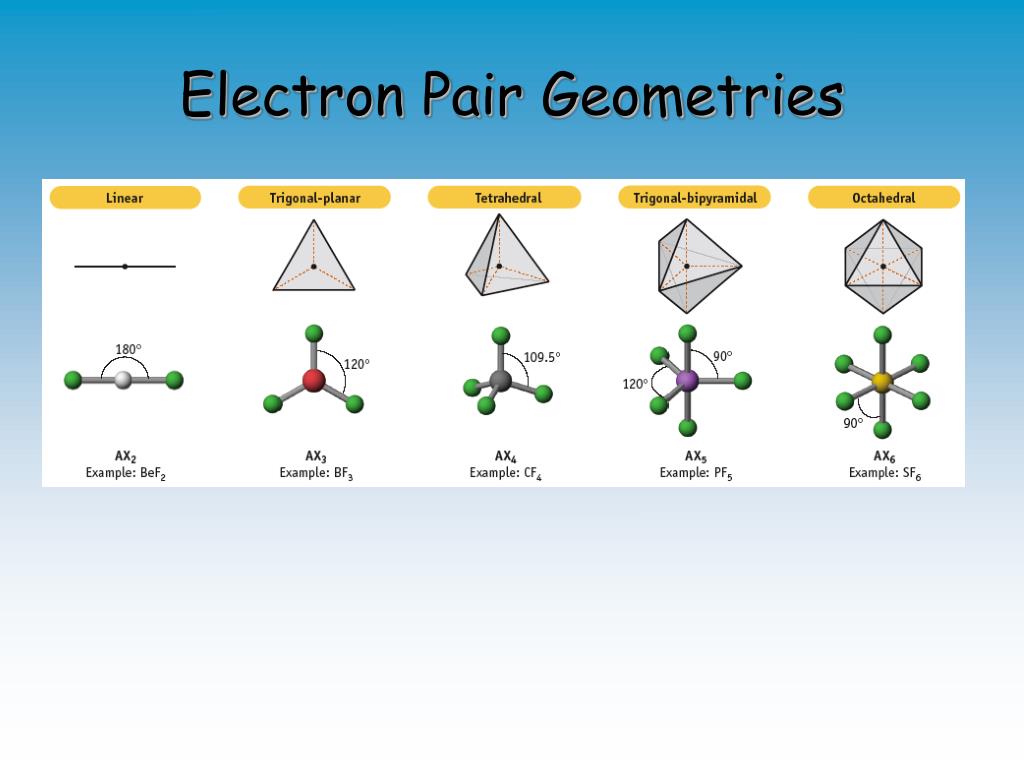
Environment, Fossil Fuels, Alternative Fuels.Gibbs Free Energy Concepts and Calculations.Calculating Standard Reaction Entropies (e.g. 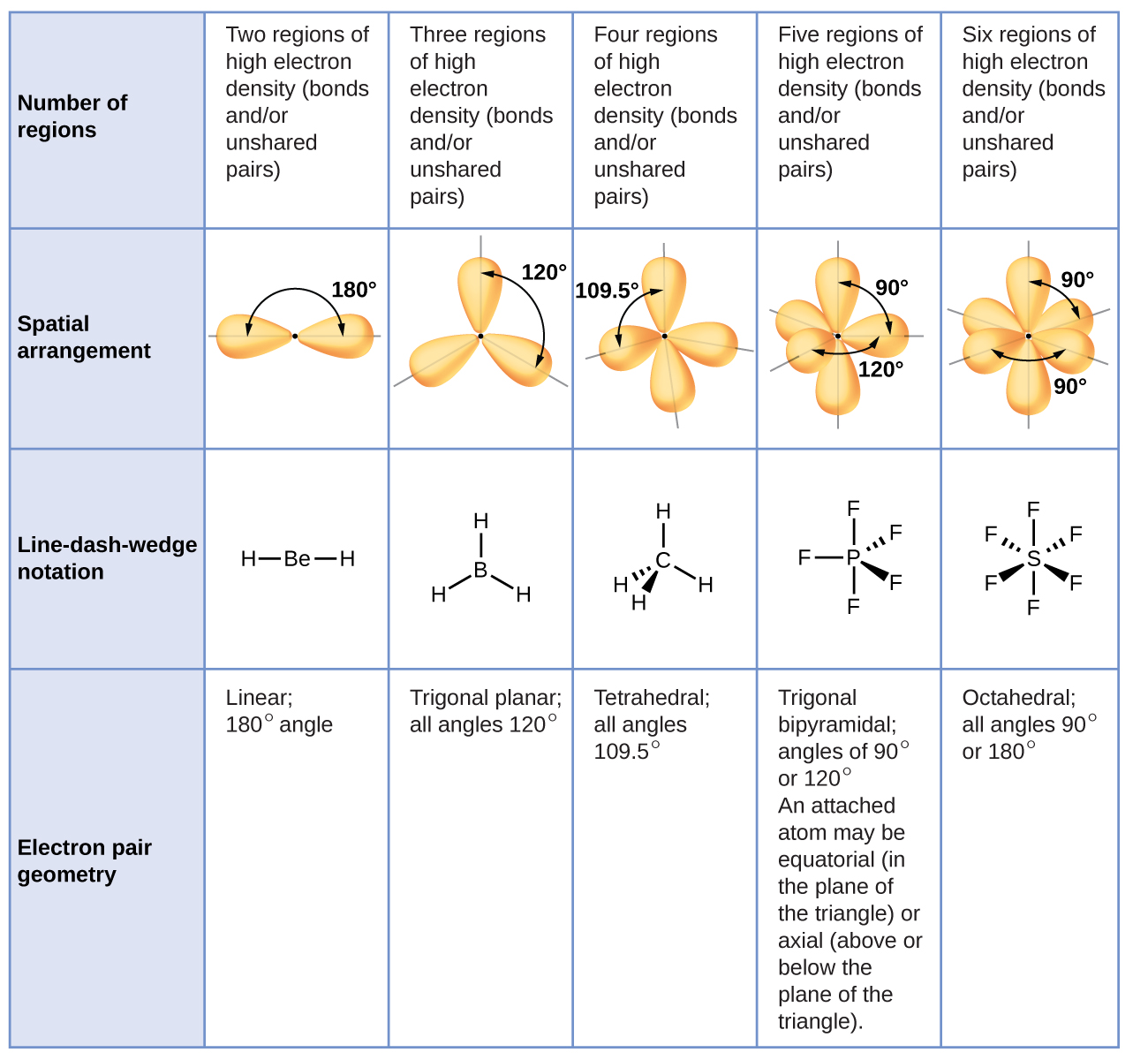
Entropy Changes Due to Changes in Volume and Temperature.Third Law of Thermodynamics (For a Unique Ground State (W=1): S -> 0 as T -> 0) and Calculations Using Boltzmann Equation for Entropy.Concepts & Calculations Using Second Law of Thermodynamics.Concepts & Calculations Using First Law of Thermodynamics.Thermodynamic Definitions (isochoric/isometric, isothermal, isobaric).Thermodynamic Systems (Open, Closed, Isolated).Heat Capacities, Calorimeters & Calorimetry Calculations.Reaction Enthalpies (e.g., Using Hess’s Law, Bond Enthalpies, Standard Enthalpies of Formation).Applying Le Chatelier's Principle to Changes in Chemical & Physical Conditions.Non-Equilibrium Conditions & The Reaction Quotient.Equilibrium Constants & Calculating Concentrations.*Biological Importance of Buffer Solutions.*Making Buffers & Calculating Buffer pH (Henderson-Hasselbalch Equation).Administrative Questions and Class Announcements.Chem 14A Uploaded Files (Worksheets, etc.).Calculating pH or pOH for Strong & Weak Acids & Bases.Acidity & Basicity Constants and The Conjugate Seesaw.Properties & Structures of Inorganic & Organic Bases.Properties & Structures of Inorganic & Organic Acids.*Molecular Orbital Theory Applied To Transition Metals.Shape, Structure, Coordination Number, Ligands.Coordination Compounds and their Biological Importance.*Molecular Orbital Theory (Bond Order, Diamagnetism, Paramagnetism).*Liquid Structure (Viscosity, Surface Tension, Liquid Crystals, Ionic Liquids).Interionic and Intermolecular Forces (Ion-Ion, Ion-Dipole, Dipole-Dipole, Dipole-Induced Dipole, Dispersion/Induced Dipole-Induced Dipole/London Forces, Hydrogen Bonding).Polarisability of Anions, The Polarizing Power of Cations.Electron Configurations for Multi-Electron Atoms.Wave Functions and s-, p-, d-, f- Orbitals.Heisenberg Indeterminacy (Uncertainty) Equation.Bohr Frequency Condition, H-Atom, Atomic Spectroscopy.Accuracy, Precision, Mole, Other Definitions.Review of Chemical & Physical Principles.Multimedia Attachments (click for details). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
